|
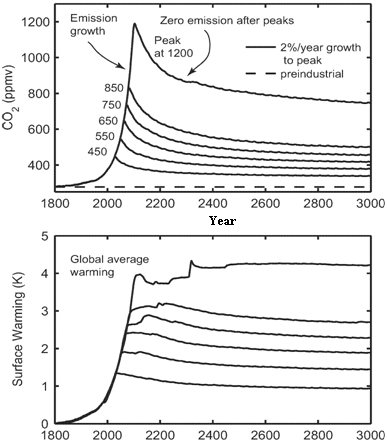
Solomon and co-authors write, “It is sometimes imagined that slow processes such as climate changes pose small risks, on . . . the assumption that a choice can be made to quickly reduce emissions and thereby reverse any harm within a few years or decades. This . . . is incorrect for carbon dioxide emissions, because of the longevity of atmospheric CO2 . . . and ocean warming.” The figure at upper right, reproduced from her paper, conveys Solomon’s point succinctly. If CO2 emissions grow by 2% per year, as they have since 2000, the concentration of the gas in the atmosphere climbs rapidly in this century. For the sake of argument, the authors assume that all emissions of CO2 suddenly cease when a “target” concentration is reached (for example, at 550 parts per million [ppm]). The level is now at 385 ppm, and was at 280 ppm before the industrial era. They wanted to see how quickly |

|
Global Methane Levels Rising Once Again Suspect melting permafrost in tundra or under sea floor
Global methane levels have remained flat for eight years since 1999 while levels of the more predominant carbon dioxide (CO2) have increased 5%. Methane is generated both by nature and by human activities, just like CO2, and its abundance has more than doubled since pre-industrial times, according to the World Meteorological Organization. So it is unwelcome news that the concentrations of methane are once again on the rise, at least since the start of 2007. In reporting observations from Australia (footnote 1), Professors M. Rigby and R. Prinn explored what may be driving the increase, and consider it likely that the source is in the Northern Hemisphere. Two distinct sources in the Arctic, one on land and the other under the sea, may be responsible for the new emissions of this greenhouse gas, and both are related to Arctic warming. It is known that wetlands release more methane to the atmosphere than any other source of the gas. Mikhail Mastepanov and colleagues were monitoring the emission of methane and CO2 from tundra soils in far northern Greenland. Usually they go home in early September, but funding in the International Polar Year of 2007-08 allowed them to stay many weeks longer. Their prolonged stay led to a discovery that in permafrost soils on land, wet tundra may be releasing a burst of methane when it freezes in the late autumn. Mastepanov and colleagues report (2) that tundra soil released as much methane when it was freezing as it did during the entire growing season. Large expanses of the continental shelf underneath the shallow margins of the Arctic Ocean just north of Siberia contain permafrost in the sediments. These sediments were once above sea level, during the glacial climate of the Ice Ages when sea level was lower. Like most freshwater wetlands, the ancient tundra also generated methane, some of which became trapped in the permafrost as the soil froze. As the sea rose, it covered the former wetlands, but the permafrost remained frozen as long as the mean annual temperature of the sediments in the shallow Arctic seas remained below 0°C. (The sea water overlying the sediments can remain unfrozen at temperatures below 0°C because of the salt content of the water lowers the freezing point.) Russian investigator Igor Semiletov reported that some of these shallow Siberian seas have extremely high concentrations of methane. He speculated that undersea sediments were warmed during the recent ice-free summers to the point that long-trapped methane was released into the water. He added that 80% of the sub-sea permafrost now was between -1°C and 0°C and susceptible to releasing methane. He reported the news at the meeting of the American Geophysical Union (AGU) last December. Both mechanisms, if confirmed, can lead to a positive feedback cycle, in which newly released methane drives further warming in the Arctic through the greenhouse effect, which then leads to more thawing of permafrost or sub-sea sediments that contain “prehistoric” methane. The Russian scientists exhibited concern, saying they saw the first signs of methane release from the sea floor, and that sub-sea sediments are “the most fragile part of the cryosphere.” CITATIONS
1. “Renewed growth of atmospheric methane” by M. Rigby and R. G. Prinn et al, Geophysical Research Letters, Vol. 35, L22805, doi:10.1029/2008GL036037, 2008.
2. “Large Tundra methane burst during onset of freezing” by Mikhail Mastepanov et al, Nature, vol. 456, 628-630, 4 December 2008. |
|
the atmosphere would recover from the buildup of CO2 from human activities if the activities were to stop: a “test case.” They explore six different targets for atmospheric CO2, getting six curves for the way that CO2 rises to a peak level, then falls off after emissions stop. Using their knowledge of atmospheric chemistry and published models, they calculated whether and how fast the level of CO2 in the air would drop as the ocean absorbs the gas, and how fast temperatures would fall as a result. Indeed, the level of CO2 in the atmosphere starts to fall when emissions cease, quickly at first, then quite slowly (top graph). But the temperature of the Earth’s surface, having climbed to a peak at the same time (lower graph), remains essentially near that peak temperature, falling much less than one degree C for hundreds of years after emissions of the gas cease. Sea level would rise at least 0.4 to 1 meter if CO2 concentrations are allowed to reach a fairly modest level of 600 ppm in this century. The seas could rise twice as much if the melting of ice sheets on land is considered. How is this possible? First, CO2 levels will not fall to pre-industrial levels. Because of the chemistry and the slow rate of mixing in the ocean, the levels fall and then plateau; in the long term the increase of CO2 in the atmosphere above the pre-industrial level will stabilize at about 40% of the peak increase that occurs when emissions stop. This is shown in the top panel of the Figure. The CO2 level in the atmosphere never falls to pre-industrial values, and it never even falls to today’s level. Then, why do temperatures not fall in the atmosphere while CO2 concentrations are falling, after the magic day when emissions stop? As the ocean warms (at first) then cools, there is a lot of inertia. The Ocean is still warming while the atmosphere is cooling, so the rate that heat flows from the air to water drops off. As the atmosphere passes less and less heat to the ocean, CO2 is still warming the atmosphere, but this warming by CO2 decreases as the amount of the gas falls. The two effects are almost in balance, and the atmosphere remains warm, within ½°C of the peak temperature that was reached. What does this portend for patterns of rainfall or drought? A “robust characteristic” of climate change in atmospheric models is an expansion of the so-called “Hadley cell,” the predominant circulation in the tropics and subtropics. That implies that the belt of dry climates now running through the latitude of Mexico and the Sahara will shift north, which means the American Southwest and the Mediterranean basin can expect drier conditions. More frequent drought has already been documented in these regions. Solomon presents a map indicating many regions (North Africa, southern Europe, west Australia) can expect 10% less rainfall in the dry season per degree of warming – and many expect 2°C of warming by the year 2050. She reminds us, “The American dust bowl was associated with an average rainfall decrease of 10% over 10 to 20 years.” Rising sea level is an irrevocable result of carbon dioxide emitted in the past, even if the emissions are totally stopped, according to the paper. The next figure, illustrates a calculation of the lower limit to possible sea level rise, depending on the peak “target” CO2 concentration. This shows the “lower limit” of sea level rise because melting of ice is not considered here. A one meter rise in sea level is a possible consequence of a target CO2 level of 550 ppm (we are now at 385 ppm); a rise of 2 meters is possible from a target of 1000 ppm. Solomon concludes that changes in the geography of the Earth’s coastlines are irreversible as many coasts and islands will be submerged. The take-home message: if CO2 emissions are stopped today, further warming and sea level rise are inevitable and irreversible. The CO2 has already been released.
CITATIONS
1. “Irreversible climate change due to carbon dioxide emissions” by Susan Solomon, G.K. Plattner, R. Knutti, and P. Friedlingstein (2009). Proceedings of the Nat. Acad. of Sciences, vol. 186, no. 6, 1704-1709, February 10, 2009. [http://www.pnas.org/cgi/doi/10.1073/pnas.0812721106]
2. “Climate Change 2007: the Physical Science Basis,” Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change (IPCC). Edited by S. Solomon et al (2007). Cambridge Univ. Press, Cambridge, UK, and New York, NY. [http://www.ipcc.ch/ipccreports/ar4-wg1.htm]
RELATED STORY: How warming has impacted ecological communities and the physical landscape on six continents - Full story |
|
Large Part of Climate Change Deemed “Irreversible” Temperatures remain elevated & stable even after carbon dioxide emissions cease |
|
Brown Clouds, Black Carbon, and “Soot” are Agent #2 in Climate Warming Second in importance after CO2: See Page 3 |
|
Some people assume that climate will revert to a manageable, less harmful state once humans stop putting greenhouse gases into the atmosphere, and that this reversal can happen in our lifetimes or that of our children. Nothing could be further from the truth, says Susan Solomon, who published a paper this month on “Irreversible Climate Change due to Carbon Dioxide Emissions” (see note 1). Solomon is the coordinating lead author of “Climate Change 2007: the Physical Science Basis” (2) of the Intergovernmental Panel on Climate Change, IPCC, and discoverer of the ozone hole over Antarctica. When carbon dioxide (CO2) emissions stop, she explains, temperatures do not fall to earlier levels, but rather remain elevated and essentially the same for centuries. Solomon maintains that the climate change expected from CO2 emissions in the first half of the 21st century is largely irreversible for 1000 years after emissions stop. In fact, sea level will continue to rise for many centuries after no more CO2 is added to the atmosphere. The reason for this unhappy consequence is that the world’s oceans have tremendous mass, and the extra heat from a warming climate and the extra carbon dioxide take a very long time indeed to be mixed into the deep ocean.
|
|
Contents |
|
CLIMATE SCIENCE FORUM Chief Editor: Michael A Fortune, Ph.D.
Email: editor (at) climate-science.org Published quarterly at Tel: (301) 754-1766 |